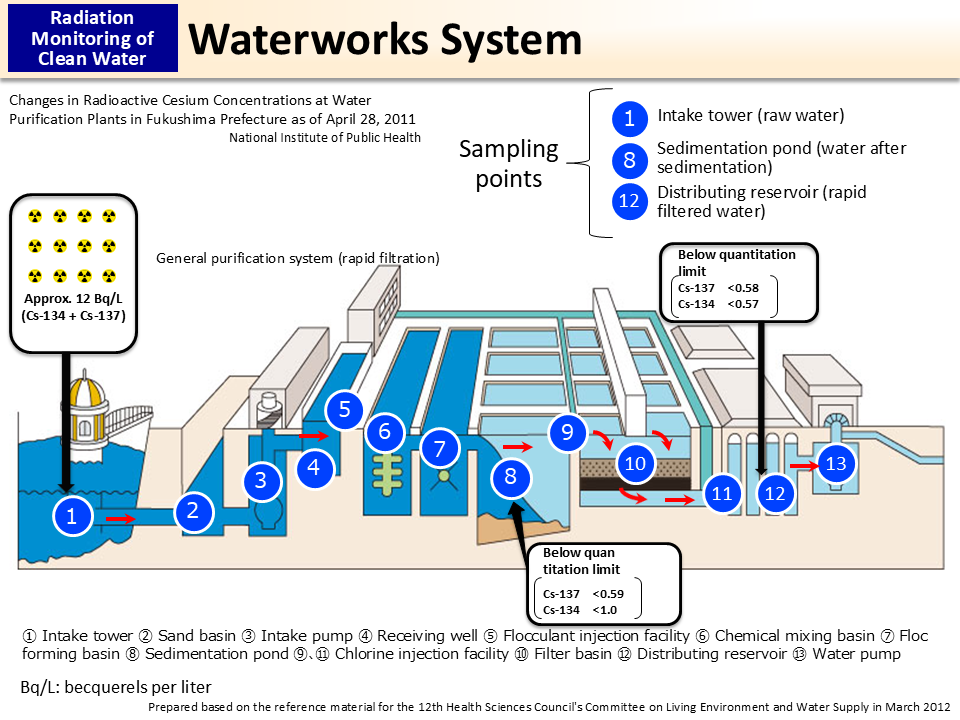
Waterworks System
This figure shows the rapid filtration method, which is generally used in Japan. In this method, chemicals are injected into raw water taken from a river or dam to cause sedimentation of mud and small particles and make them into big chunks called “floc.” Tap water is created by filtering the clear upper portion of such water.
Cesium has the property to be easily adsorbed into soil and mud (p.42 of Vol. 2, “Behavior of Radioactive Cesium”). Therefore, when water is separated from floc, cesium tends to gather around the floc, which is a chunk of soil and mud. Additionally, tap water is created using the clear upper portion of the water in plant basins. Therefore, this mechanism leaves little chance for cesium to be mixed into tap water.
In the pattern diagram above, radioactive cesium concentrations (Bq/L) actually measured at a water purification plant in Fukushima Prefecture as of April 28, 2011, are indicated at points where measurement was conducted. Radioactive cesium concentration, which was initially approx. 12 Bq/L at the intake tower, decreased to below the quantitation limit in the end when being pumped out from the distribution reservoir. As 1 liter of water weighs approx. 1 kg, it can be found that the concentration was far below 200 Bq/kg, which was the allowable limit for radioactive cesium in tap water publicized by the Ministry of Health, Labour and Welfare (MHLW) in March 2011, and also far below 10 Bq/kg, which is specified in the new standards for radioactive materials in tap water publicized in March 2012 (p.55 of Vol. 2, “Standard Limits Applied from April 2012”).
- Included in this reference material on March 31, 2015
- Updated on March 31, 2019