Health & Chemicals
APPROACH TO CHEMICALS SUSPECTED OF HAVING ENDOCRINE DISRUPTING EFFECTS
November 2002
MINISTRY OF THE ENVIRONMENT, GOVERNMENT OF JAPAN
This paper describes an approach to chemicals suspected of having endocrine disrupting effects currently taken by the Ministry of the Environment of Japan (MoE). Although MoE considers that it is still too early to decide a testing/assessing scheme of endocrine disrupting substances, we think it might be useful to present MoE's approach, in particular on mammals, as a thought starter for further consideration of the testing and assessing strategy at the OECD level.
INTRODUCTION
Ministry of the Environment of Japan (MoE) has released a document entitled "Strategic Programs on Environmental Endocrine Disrupters (SPEED'98)" on May 1998 and listed 67 substances as "chemicals suspected of having endocrine disrupting effects". The Priority List was revised in November 2000 due to a deletion of two substances (styrene dimer/trimer and n-butylbenzene) of which estimated risks were low. Thus, 65 chemicals are listed at present (See ANNEX 1).
MoE selected 12 chemicals1) in the Priority List, as high-priority chemicals, in the year 2000 and initiated their hazard assessments etc. In the year 2001 and 2002, additional 8 chemicals2) were selected and their hazard assessment etc. began, respectively.
SCREENING AND TESTING
The present status of the screening and testing for identifying possible endocrine disrupting effects of the above-mentioned 28 chemicals are described below (See also Annex 2):
* Mammals
1. In vivo study
(1) One-Generation Study in Rodents
- A pilot study of the One-Generation Study was completed for 10 of the 12 substances1) except for two alkylphenols. The results were announced on 14th June 2002. It should be noted that as for di-n-butylphthalate, additional tests have currently been conducted.
- Since two alkylphenols (4-octylphenol and nonylphenol) were suspected of having an endocrine disrupting effect, pilot studies using EE as a positive control were conducted and completed. Based on the results of the pilot studies, the One-Generation Study was designed and is being conducted for the two chemicals.
(iii) The pilot study of the One-Generation Study is now being conducted concerning 8 chemicals2).
(iv) Study diagram
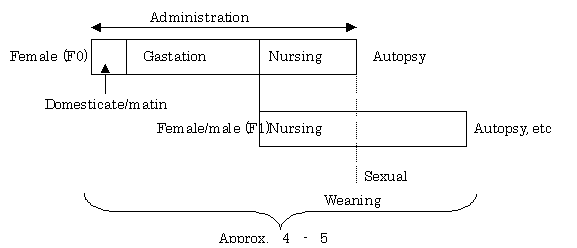
(2) Uterotrophic assay
Tests have been completed for 12 substances1),3).
(3) Hershberger assay
Tests have been completed for 12 substances1),3).
(4) Enhanced OECD TG 407: Repeated Dose 28-day Oral Toxicity Study
Based on the results that METI4), MHLW5) and MAFF6) validate the test substances requested by OECD, research and developmental studies are being conducted.
2. In vitro study
(1) Human estrogen receptor (hER alpha and hER beta) binding assay and E-screen assay
The study has been completed for 12 substances1),3).
(2) Human androgen receptor (hAR) reporter gene assay and Rat androgen receptor (rAR) binding assay
The study has been completed for 12 substances1),3).
(3) Human thyroid hormone receptor (hTR alpha and hTR beta) yeast assay
The study has been completed for 12 substances1),3) .
3. DNA microarray
Developed and validated methods for risk assessment of EDs using DNA microarray technology could provide alternatives to traditional animal bioassays. E2 (control) and 7 substances (tributyltin, 4-octylphenol, nonylphenol, di-n-butylphthalate, octachlorostyrene, benzophenone and dicyclohexylphthalate ; test chemicals) were examined gene expression regulation by the test chemicals on human cell lines and mouse cell lines. In the case of human cell lines, a microarray comprising about 8,400 human genes was used for the test. A statistically significant change in expression was observed for about 1,000 genes in the microarray. In the case of mice, about 1,500 of approximately 9,000 genes were identified. We are currently preparing microarrays (ED chip) including selected expression regulated genes by EDs, both for humans and mice. The goal includes establishment of test methods, classification of expression profiles, and construction of databases on individual EDs using the ED chip.
* Ecosystem
1. Fish (Medaka used in all cases) (See Annex 3)
(1) Screening
(i) Vitellogenin assay The study has been completed for 12 substances1),3) and 8 substances2).
(ii) Partial life cycle test
The study has been completed for 12 substances1),3). The results and the guideline are described in the attached papers "Results of Assay and Tests in Evaluation of the Endocrine Disrupting Activeties in Fish (Medaka)" and "The medaka partial life-cycle test guideline".
(iii) Development of FLF/d-rR Medaka
Early life stage study using Medaka (FLF/d-rR) in which genetic sex can be distinguished in such a simple manner as observing its leucophore or its color of body has been developed.
(2) Testing
- Full life cycle test -
The study has been completed for 3 substances (nonylphenol, 4-octylphenol and di-n-butylphthalate). The result of nonylphenol was published as a risk assessment report on August 2001, and the results of the other two substances were announced3). The results and the guideline are described in the attached papers "Results of Assay and Tests in Evaluation of the Endocrine Disrupting Activeties in Fish (Medaka)" and "The medaka full life-cycle test guideline".
(3) In vitro study
(i) Medaka estrogen receptor (mER alpha and mER beta) binding assay and Medaka estrogen receptor (mER alpha and mER beta) reporter gene assay
The study has been completed for 12 substances1),3).
(ii) Medaka androgen receptor (mAR) reporter gene assay
The study has been completed for 12 substances1),3).
(4) Others (The study on the mechanism of sexual differentiation, DNA chip and DMY)
In order to make clear the effects and their mechanism of the EDs to the differentiation of sex concerning Medaka with utilizing the genetic technology, some studies have been conducted as follows:
- Cloning a series of genes associated with regulating the differentiation of sex concerning Medaka
- The sex-determining gene of Medaka, DMY (Y-specific DM-domain gene) has been first found in non-mammalian vertebrates. The report was published in the science magazine "Nature". (Matsuda et al. DMY is a Y-specific DM-domain gene required for male development in the medaka fish. Nature 417, 559-563 (2002))
- Preparation of a gene, which appears in a period of differentiation of sex in order to develop a DNA chip containing a series of genes associated with the differentiation of sex concerning Medaka
2. Birds
(1) Screening
(i) Vitellogenin assay
The study has been completed for 12 substances1) and 8 substances2). The vitellogenin ELISA assay system was established using a quail antigen. This method is very specific for detecting endocrine disruption in birds. The sensitivity of the ELISA is very high (comparable to those for fish vitellogenin).
(ii) Kuro-Aka (Cloacal) protrusion test
The cloacal protrusion of Japanese quail is a target organ of circulating androgens and its androgenic stimulation is easily estimated non-invasively by measuring its area. Using this structure as an endpoint, anti-androgenic activity of test chemicals can be estimated. The test system is currently being established and a pilot test is undertaken.
(iii) Sex reversal test
Japanese quail has a strain which shows a sex-linked plumage color mutation. Checking plumage color and detecting gonadal abnormality, effects of administered chemicals on gonadal development can be detected before hatching. A pilot test is currently under examination using positive standard substances.
(2) Development of testing
In order to develop a method of Two-Generation Reproductive Toxicity Study, basic research of (1) sex differentiation and growth, (2) fertility, (3) development, and (4) central nervous system function/behavior to be conducted.
(3) In vitro study
Japanese quail estrogen receptor (qER alpha and qER beta) binding assay
The study has been completed for 12 substances1),3).
3. Amphibians
(1) Screening
(i) Metamorphosis assay
Frog metamorphosis assay using tadpole for detecting thyroid hormone-like effect is under development. A pilot study with Xenopus laeviswas completed successfully. The next step is to participate in the international ring test.
Basic research using native Japanese species (Rana rugosa) is in progress.
(ii) Vitellogenin assay
Currently being developed.
(2) In vitro study
Estrogen receptor binding assay
Currently being developed.
4. Invertabrates
(1) Screening and testing
Currently being developed.
PROTOCOLS AND TEST RESULTS
The present status of the protocols and the results in brief of the tests, which were recently taken by the Ministry of the Environment of Japan (MoE) and required by the experts who participated in EDTA 6th (June 24-25 Tokyo, Japan).
*Mammals
- Tested Chemicals
Tributyltin, di-n-Butylphthalate, Octachlorostyrene, Benzophenone, di-Cyclohexylphthalate, di-(2-Ethylhexyl)phthalate, Butylbenzylphthalate, di-Ethylphthalate, di-(2-Ethylhexyl)adipate and Triphenyltin
- Protocols of in vivo study
See ANNEX 4-1 to 4-10: "Summary of Protocol" of 10 Chemicals.
- Results of in vivo and in vitro study
See ANNEX 5-1 to 5-10: "Results of Assays and Tests in Evaluation of the Endocrine Disrupting Activities in Rodent".
The results in detail are described in the attachment of "Summary of Results"
- Ecosystem -Fish (Medaka used in all cases)-
*Ecosystem -Fish(Medaka used in all cases)-
- Tested Chemicals
4-t-Octylphenol, di-n-Butylphthalate, di-(2-Ethylhexyl)phthalate, di-Cyclohexylphthalate, di-Ethylphthalate, Butylbenzylphthalate, di-(2-Ethylhexyl)adipate, Tributyltin, Benzophenone and Octachlorostyrene
- Guideline of screening and testing
See ANNEX 6-1:for the Guideline of Partial Life-Cycle Test and Full Life-Cycle Test:
"The medaka (Oryzias latipes) Partial Life-Cycle Test Guideline".
See ANNEX 6-2:for the Guideline of Full Life-Cycle Test and Full Life-Cycle Test:
"The medaka (Oryzias latipes) Full Life-Cycle Test Guideline".
- Results of in vivo and in vitro study
See ANNEX 7: "Results of Assay and Tests in Evaluation of the Endocrine Disrupting Activities in Fish(Medaka)".
Note 1: 12 chemicals selected in 2000
Tributyltin, 4-octylphenol, nonylphenol, di-n-butylphthalate, octachlorostyrene, benzophenone, di-cyclohexylphthalate, di-(2-ethylhexyl)phthalate, butylbenzylphthalate, di-ethylphthalate, di-(2-ethylhexyl)adipate and triphenyltin
Note 2: Each 8 chemicals selected in 2001 and 2002
Pentachlorophenol, amitrole, bisphenol A, 2,4-dichlorophenol, 4-nitrotoluene, di-pentylphthalate, di-hexylphthalate and di-propylphthalate in 2001
Hexachlorobenzen, Hexachlorocyclohexane, Chlordane, Oxychlordane, trans-Nonachlor, DDT, DDE, DDD in 2002
Note 3: The results were announced on 14th June 2002.
Note 4: METI : Ministry of Economy, Trade and Industry, Japan
Note 5: MHLW : Ministry of Health, Labor and Welfare, Japan
Note 6: MAFF : Ministry of Agriculture, Forestry and Fisheries, Japan